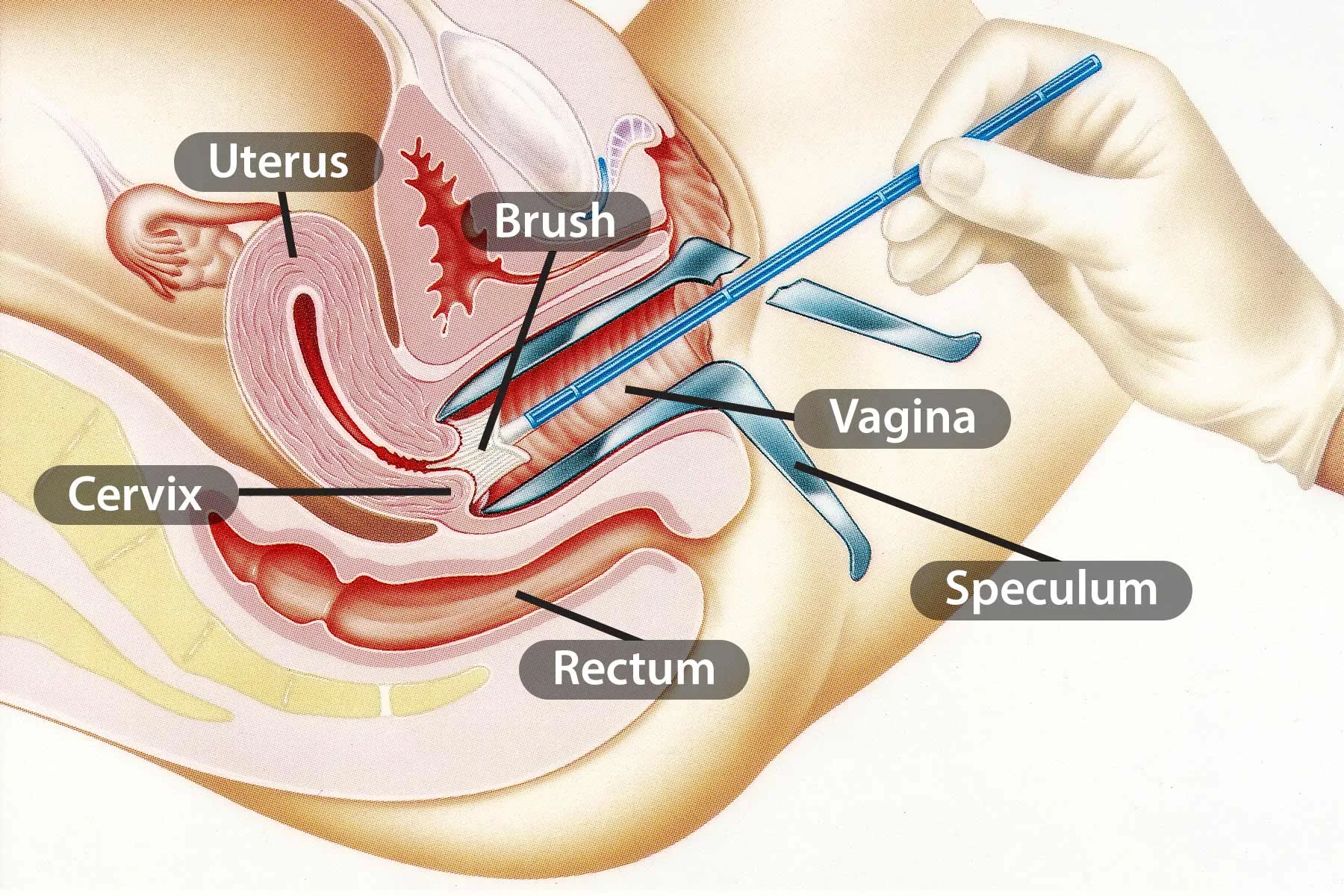
Pap smear is a cervical cancer screening test that involves collecting cells from the cervix to detect abnormal cellular changes that could indicate precancerous conditions or cervical cancer. The Pap smear, also known as a Pap test or cervical cytology, is performed during a pelvic examination where a healthcare provider uses a speculum to visualize the cervix and collects cells using a brush or spatula for microscopic analysis. At the population level, regular Pap smear screening has reduced cervical cancer incidence and mortality by over 70% in countries with organized screening programs, making it one of the most successful cancer prevention strategies in modern medicine.
Pap Smear | |
|---|---|
| Category | Women’s Health, Cancer Screening, Gynecology |
| Research Fields | Gynecology, Oncology, Pathology, Public Health, Epidemiology |
| Also known as: | Pap test, cervical cytology, cervical smear |
| Pronunciation: | pap smeer |
| Recommended age start: | 21 years old |
| Screening frequency: | Every 3 years (ages 21-29) |
| Co-testing frequency: | Every 5 years (ages 30-65 with HPV test) |
| Sensitivity: | 55-85% for detecting cervical abnormalities |
| Cancer reduction: | 70-80% reduction in cervical cancer incidence |
| Sources: | ACOG, CDC, NEJM, WHO |
Current screening guidelines recommend that women begin Pap smear testing at age 21 regardless of sexual activity onset, with screening every 3 years for women ages 21-29, and co-testing with HPV testing every 5 years for women ages 30-65. The test has a sensitivity of 55-85% for detecting cervical abnormalities, meaning it may miss some abnormal cells, which is why regular screening intervals are crucial for optimal cancer prevention effectiveness.
Approximately 13,000 new cases of cervical cancer are diagnosed annually in the United States, with over 4,000 deaths, representing a dramatic decline from pre-screening era rates when cervical cancer was a leading cause of cancer death in women. Understanding Pap smear screening encompasses knowledge of preparation requirements, procedure expectations, result interpretation, and follow-up care recommendations for abnormal findings.
History
Pre-Screening Era
Before the development of cervical screening, cervical cancer was one of the leading causes of cancer death among women worldwide. The disease typically went undetected until advanced stages, when treatment options were limited and survival rates were poor.
1928: George Papanicolaou’s Discovery
Greek-American physician George Papanicolaou first observed that cervical cells could be collected and examined microscopically to detect cancer. His initial observations were published in a paper titled “New Cancer Diagnosis,” though the medical community initially showed limited interest in his findings.
1940s: Clinical Development
Papanicolaou refined his technique and, along with gynecologist Herbert Traut, published detailed methods for cervical cytology in their 1943 book “Diagnosis of Uterine Cancer by the Vaginal Smear.” This work established the foundation for widespread clinical application.
1950s: Mass Screening Programs
The first organized cervical cancer screening programs began in the 1950s, initially in developed countries. These programs demonstrated dramatic reductions in cervical cancer incidence and mortality, validating the effectiveness of population-based screening.
1960s-1970s: Widespread Adoption
Pap smear screening became routine gynecological care in most developed countries during this period. Standardized laboratory procedures, quality control measures, and training programs for cytotechnologists were established to ensure test accuracy and reliability.
1980s: Bethesda System
The Bethesda System for reporting cervical cytology was introduced in 1988, providing standardized terminology for Pap smear results. This system improved communication between laboratories and clinicians and standardized follow-up recommendations.
1990s: HPV Discovery
The discovery that human papillomavirus (HPV) causes virtually all cervical cancers revolutionized understanding of cervical cancer etiology and led to development of HPV testing as an adjunct to Pap smears.
2000s-Present: Integrated Screening
Modern cervical cancer screening combines Pap smears with HPV testing (co-testing) and has evolved to include HPV vaccination as primary prevention. Liquid-based cytology has largely replaced conventional Pap smears, improving specimen quality and enabling HPV testing from the same sample.
Procedure and Technique
Pre-Procedure Preparation
Optimal Pap smear preparation includes scheduling the test for mid-cycle (avoiding menstruation), abstaining from sexual intercourse for 24-48 hours before the test, avoiding douching or vaginal medications for 2-3 days, and ensuring the patient isn’t using vaginal lubricants or spermicides.
Examination Setup
The procedure is performed during a pelvic examination with the patient in lithotomy position. A speculum is inserted into the vagina to visualize the cervix, and adequate lighting is essential for proper specimen collection. The cervix is inspected for visible abnormalities before cell collection.
Cell Collection Methods
Modern Pap smears use liquid-based cytology where cells are collected using a plastic spatula and cytobrush, then suspended in liquid preservative medium rather than smeared directly onto slides. This method improves specimen adequacy and allows for additional testing including HPV detection.
Specimen Processing
Collected specimens are transported to cytology laboratories where automated systems prepare thin-layer slides for microscopic examination. Cytotechnologists and pathologists examine slides for cellular abnormalities, with computer-assisted screening increasingly used to improve accuracy and efficiency.

Cervical Cytology and Pathology
Normal Cervical Cells
Normal cervical epithelium consists of squamous cells from the ectocervix and glandular cells from the endocervix. The transformation zone, where squamous and glandular epithelia meet, is the most important area for screening as it’s where most cervical cancers develop.
Cellular Changes and Progression
Cervical cancer typically develops through a progression of increasingly severe cellular abnormalities: normal epithelium → low-grade squamous intraepithelial lesion (LSIL) → high-grade squamous intraepithelial lesion (HSIL) → invasive cancer. This progression usually takes 10-20 years, providing ample opportunity for detection and intervention.
HPV-Related Changes
Human papillomavirus infection causes characteristic cellular changes visible on Pap smears, including koilocytosis (cells with enlarged nuclei and clear halos), dyskeratosis, and nuclear atypia. These changes indicate HPV infection and varying degrees of cellular transformation.
Bethesda System Classification
The Bethesda System categorizes Pap smear results as: Normal/Negative, ASC-US (atypical squamous cells of undetermined significance), ASC-H (atypical squamous cells, cannot exclude HSIL), LSIL, HSIL, and invasive cancer. Each category has specific follow-up recommendations based on risk stratification.
Screening Guidelines and Recommendations
Age-Based Screening
Current guidelines recommend starting Pap smear screening at age 21 regardless of sexual activity onset, reflecting the low risk of cervical cancer in younger women and potential harms of early screening. Screening before age 21 is not recommended even for sexually active teenagers.
Screening Intervals
Women ages 21-29 should have Pap smears every 3 years (cytology alone), while women ages 30-65 can choose Pap smears every 3 years or co-testing (Pap plus HPV) every 5 years. The longer interval for co-testing reflects the improved sensitivity of combined testing.
Discontinuation Guidelines
Screening can be discontinued at age 65 for women with adequate prior screening and no history of significant abnormalities. Women who have had total hysterectomy for benign conditions don’t require routine screening unless they have a history of cervical abnormalities.
Special Populations
Immunocompromised women, those with HIV infection, and women with history of cervical abnormalities may require more frequent screening. Specialized screening protocols address the increased cancer risk in these populations.
HPV Co-Testing
HPV Testing Technology
HPV testing detects high-risk HPV types that cause cervical cancer, most commonly using DNA or RNA amplification techniques. FDA-approved tests can identify specific HPV types or provide pooled results for high-risk types, with some tests specifically identifying HPV 16 and 18.
Co-Testing Benefits
Combining Pap smears with HPV testing increases sensitivity for detecting cervical abnormalities to over 95%, compared to 55-85% for Pap smears alone. Co-testing also has excellent negative predictive value, meaning negative results provide strong reassurance against current or near-term cancer risk.
HPV Primary Screening
Some guidelines now support HPV testing alone as primary screening for women over 25-30, with Pap smears used for triage of HPV-positive women. This approach may be more sensitive than cytology-based screening and could allow longer screening intervals.
Risk Stratification
HPV genotyping allows risk stratification, with HPV 16 and 18 conferring highest cancer risk and requiring immediate colposcopy regardless of cytology results. Other high-risk HPV types are managed based on concurrent cytology findings and patient risk factors.
Abnormal Results and Follow-Up
ASC-US Management
Atypical squamous cells of undetermined significance (ASC-US) is the most common abnormal Pap result, found in 3-5% of screens. Management options include immediate HPV testing, repeat cytology in 6-12 months, or colposcopy, with HPV triage being the preferred approach for women over 25.
Low-Grade Lesions (LSIL)
LSIL represents HPV infection with mild cellular changes that often resolve spontaneously. Management typically involves either immediate colposcopy or repeat co-testing in 12 months, with colposcopy reserved for persistent abnormalities or high-risk HPV positivity.
High-Grade Lesions (HSIL)
HSIL indicates more severe cellular abnormalities with higher cancer risk, requiring immediate colposcopy and usually treatment with excisional procedures like LEEP (loop electrosurgical excision procedure) or cold knife conization to remove abnormal tissue.
Colposcopy and Biopsy
Colposcopy involves magnified examination of the cervix with acetic acid application to identify abnormal areas for targeted biopsy. Colposcopy-directed biopsies provide definitive diagnosis and guide treatment decisions based on the severity of cervical intraepithelial neoplasia (CIN).
Quality Assurance and Limitations
Laboratory Quality Control
Pap smear accuracy depends on rigorous laboratory quality control including regular proficiency testing for cytotechnologists and pathologists, automated screening systems, and correlation with histologic follow-up results. CLIA regulations ensure standardized laboratory practices.
Specimen Adequacy
Adequate specimens must contain sufficient squamous cells and endocervical/transformation zone cells for proper evaluation. Unsatisfactory specimens require repeat collection, while specimens lacking endocervical cells may need earlier repeat screening.
False Negatives and Positives
Pap smears have inherent limitations including false negative rates of 15-50% for single tests, primarily due to sampling errors, laboratory interpretation issues, and obscuring factors like inflammation or blood. False positives can lead to unnecessary anxiety and procedures.
Provider Training
Proper specimen collection requires adequate training in pelvic examination techniques, speculum use, and sampling methods. Inadequate provider technique is a significant cause of specimen inadequacy and false negative results.
Global Perspectives and Access
Developed vs. Developing Countries
Cervical cancer screening availability varies dramatically worldwide, with organized programs common in developed countries but limited access in many low- and middle-income countries where cervical cancer remains a leading cause of cancer death among women.
Alternative Screening Methods
In resource-limited settings, alternative screening methods include visual inspection with acetic acid (VIA), HPV testing using point-of-care devices, and self-collected HPV samples. These approaches can be more feasible than conventional Pap smears in areas lacking cytology infrastructure.
WHO Elimination Strategy
The World Health Organization has set goals to eliminate cervical cancer through 90% HPV vaccination coverage, 70% screening coverage, and 90% treatment of pre-cancerous lesions. Achieving these targets could prevent millions of cancer cases globally.
Economic Considerations
Cost-effectiveness analyses consistently show cervical cancer screening to be among the most cost-effective health interventions. However, implementing screening programs requires significant healthcare infrastructure investment, particularly in cytology laboratory capacity.
Technological Advances
Liquid-Based Cytology
Liquid-based cytology has largely replaced conventional Pap smears, offering improved specimen adequacy, clearer microscopic appearance, and the ability to perform HPV testing from the same sample. Automated processing systems further improve consistency and efficiency.
Computer-Assisted Screening
Automated screening systems use computer algorithms to identify potentially abnormal cells, allowing cytotechnologists to focus on suspicious areas and improving screening efficiency. These systems can also flag specimens requiring pathologist review.
Biomarker Development
Emerging biomarkers including p16/Ki-67 dual staining, methylation markers, and microRNA signatures may improve cervical cancer screening accuracy and risk stratification beyond current cytology and HPV testing approaches.
Artificial Intelligence
AI-powered systems are being developed to interpret cervical cytology and identify abnormal cells with accuracy comparable to human cytotechnologists. AI applications in cervical screening may help address cytotechnologist shortages and improve screening access.
Future Directions
HPV Vaccination Impact
Widespread HPV vaccination is expected to dramatically reduce cervical cancer incidence over the coming decades, potentially allowing less frequent screening or different screening strategies for vaccinated populations. Early data from vaccinated cohorts shows significant reductions in cervical abnormalities.
Precision Screening
Future screening may incorporate individual risk factors including HPV vaccination status, genetic markers, immune status, and lifestyle factors to personalize screening intervals and methods. Risk-based approaches could optimize benefits while minimizing harms.
Self-Collection Methods
Self-collected samples for HPV testing show promise for increasing screening participation, particularly among women who don’t regularly access healthcare. Validation studies suggest adequate accuracy for high-risk HPV detection using self-collected specimens.
Frequently Asked Questions
When should I start getting Pap smears?
Current guidelines recommend starting Pap smear screening at age 21, regardless of when you became sexually active. Screening before age 21 is not recommended because cervical cancer is extremely rare in teenagers and early screening may cause unnecessary anxiety and procedures.
How often do I need a Pap smear?
Women ages 21-29 should have Pap smears every 3 years. Women ages 30-65 can choose Pap smears every 3 years or co-testing (Pap plus HPV test) every 5 years. Your healthcare provider can help determine the best schedule based on your individual risk factors.
What should I do to prepare for a Pap smear?
Avoid sexual intercourse, douching, and vaginal medications for 24-48 hours before your test. Try to schedule your appointment when you’re not menstruating, ideally mid-cycle. You can take an over-the-counter pain reliever before the appointment if you’re concerned about discomfort.
What does an abnormal Pap smear mean?
Abnormal Pap results don’t necessarily mean you have cancer. Most abnormal results indicate HPV infection or mild cellular changes that often resolve on their own. Your healthcare provider will explain your specific results and recommend appropriate follow-up, which might include repeat testing or colposcopy.
Can I get a Pap smear if I’m a virgin?
Yes, but screening recommendations are based on age rather than sexual activity. If you’re under 21, screening typically isn’t recommended regardless of sexual activity status. If you’re over 21 and have never been sexually active, discuss with your healthcare provider whether screening is appropriate for your situation.
Is the Pap smear procedure painful?
Most women experience only mild discomfort during the procedure, which typically lasts just a few minutes. The speculum insertion and cell collection may cause brief cramping or pressure. Taking deep breaths and relaxing your muscles can help minimize discomfort.
When can I stop getting Pap smears?
Women can generally stop routine screening at age 65 if they’ve had adequate prior screening with normal results and no history of significant cervical abnormalities. Women who’ve had total hysterectomy for benign conditions typically don’t need continued screening unless they have a history of cervical problems.